Certificate: | NMPA, CE, WHO EUL, FDA EUA |
Pack size: | 48 tests/kit, 96 tests/kit |
Principles: | The real-time PCR technique can detect ORF1ab gene at FAM channel, N gene at JOE channel, E gene at ROX channel and internal reference gene at Cy5 channel |
Product Application: | It has obtained NMPA, CE, FDA EUAand WHO EUL approval, has supported many countries and regions abroad for epidemic prevention and control, and has been deeply recognized in terms of product quality and after-sales service. This product can be used for COVID-19 nucleic acid test and guarding against both human carriers and object carriers, so as to minimize the unpredictable public health impact caused by missed cases and false testing, and reduce the resources consumption caused by repeated detection of the laboratory. |

Single-tube testing of 3 targets, independently developed and designed, covering known variants, and mutual verification of 3 target results for coping with the risk of frequent virus mutation
Verification of mutation sites carried by the latest known variant in the world has been completed. Hence, there will be no off-target or missed case.
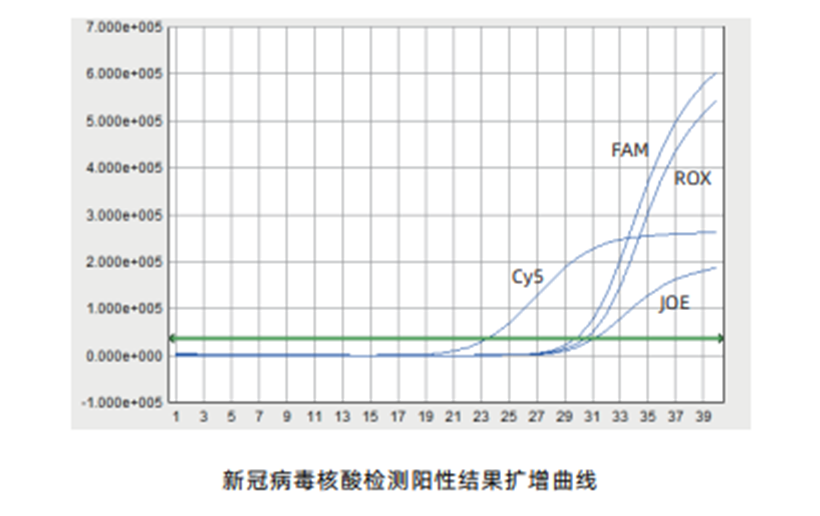
Sensitivity up to 300 copies/mL, high amplification curve signal value, and easier interpretation of results
Applicable to common four-channel PCR instument
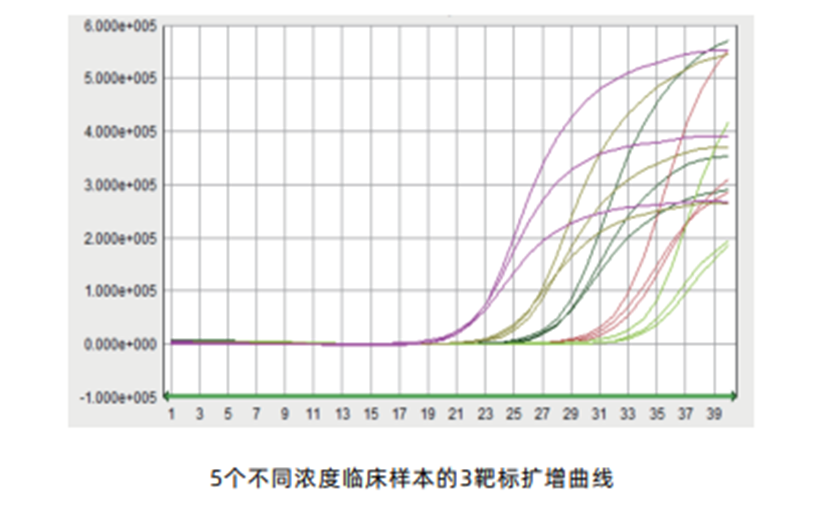
Overall clinical coincidence rate up to 98.48%, outstanding performance in user inter-lab quality evaluation and highly recognized quality
The UNG enzyme and dUTP contamination control system is used for reducing contamination of amplified products and avoid false positive results.
Internal reference is used for quality control to prevent false negative results, and is suitable for both human and object.