Enhancement solution NMPA: | STXB No. 20190302 |
| Sample Release Reagent NMPA: | STXB No. 20190301 |
Pack size: | 24 tests/kit |
Sample Type: | The serum samples can be stored at 2~8°C for not more than 7 days and at -20°C for not more than 2 years |
| Clinical significance: | By detecting the number or structural changes of oligosaccharide chains in human cells, we can understand the risk of liver cancer, so as to achieve early detection, diagnosis and treatment. |
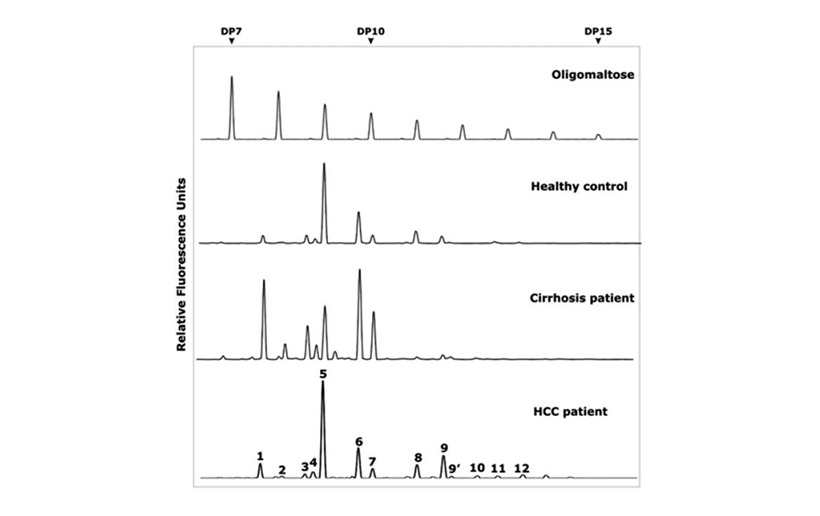
We combine the traditional biochemical specific reaction with the high sensitivity fluorescence labeling, and introduce the microelectrophoresis high separation technique to realize simple high throughput glycoprotein oligosaccharide chain fingerprint analysis of trace amount (2μl serum) under the normal biochemical laboratory conditions.
The sensitivity and specificity of the clinical registration data were 83.61% and 87.84%, respectively. Compared with the serum marker AFP of HCC , when AFP<200 and <400 were taken as the reference value of the negative result, the detection rate of AFP negative HCC can be increased by about 85%. The clinical sensitivity of G-Test and AFP combined detection is 93%.

At present, serological markers combined with imaging technology are commonly used for detection of HCC in clinical practice. However, the sensitivity and accuracy of imaging technology for lesions smaller than 1 cm are relatively low, and it is difficult to distinguish the benign lesions from the malignant lesions with the imaging technology. G-Test can meet requirements for screening test of early HCC in high-risk groups of liver cancer such as chronic hepatitis B, hepatic fibrosis and hepatic cirrhosis, and finding out the genuinely high-risk populations for more effective HCC screening follow-up.
The G-Test is applicable to early warning and screening of hepatoma risk in people infected with hepatitis B virus, auxiliary diagnosis of primary hepatocellular carcinoma in patients infected with chronic hepatitis B virus and suspected of hepatoma, and can also be used to evaluate the therapeutic effect of treatment for patients with primary hepatoma caused by hepatitis B virus and to monitor the recurrence of HCC after treatment/operation. G-Test aims to improve the accuracy of the closed loop of HCC diagnosis and treatment, make up for the deficiency of the closed loop of diagnosis and treatment, and expand the application scope of the closed loop of diagnosis and treatment.
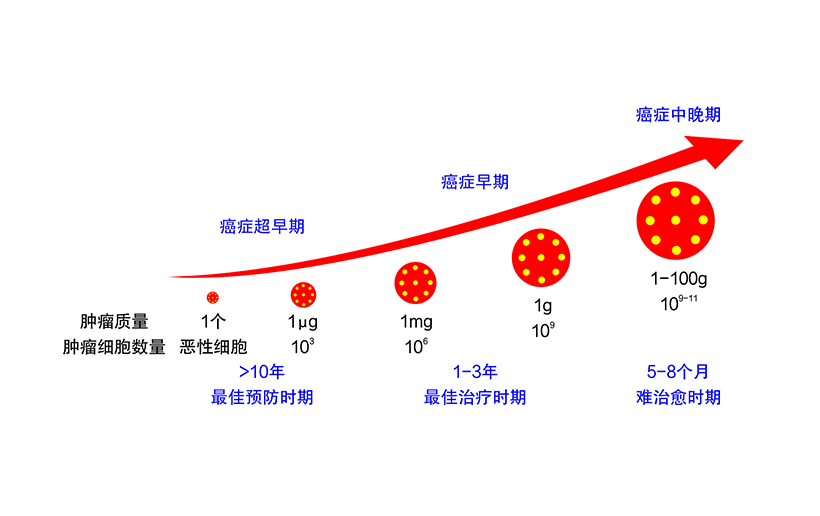
The change of glucose chain is closely related to the occurrence and development of HCC. The oligosaccharide chain detection of G-Test hepatocellular carcinoma is based on the principle of close correlation between glycomics and disease. With glucose chain map used for distinguishing the status of liver disease and liver-related diseases for early detection of HCC, the early warning of HCC can be given 2 years in advance. This has an important clinical significance for early diagnosis of HCC.